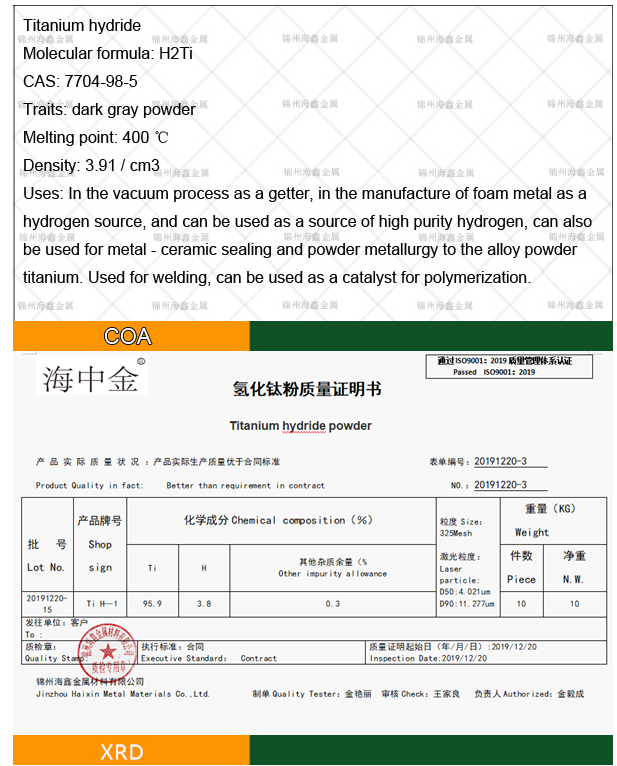

Chinese name: Titanium Dihydroxide
English name: TITANIUM HYDRIDE;
CAS number: 7704-98-5;
Melting point: 400 ℃
Water solubility: insoluble in water
Density: 3.91g/cm3
Appearance: Dark gray powder or crystal
chemical formula: TiH2
Molecular weight: 49.89


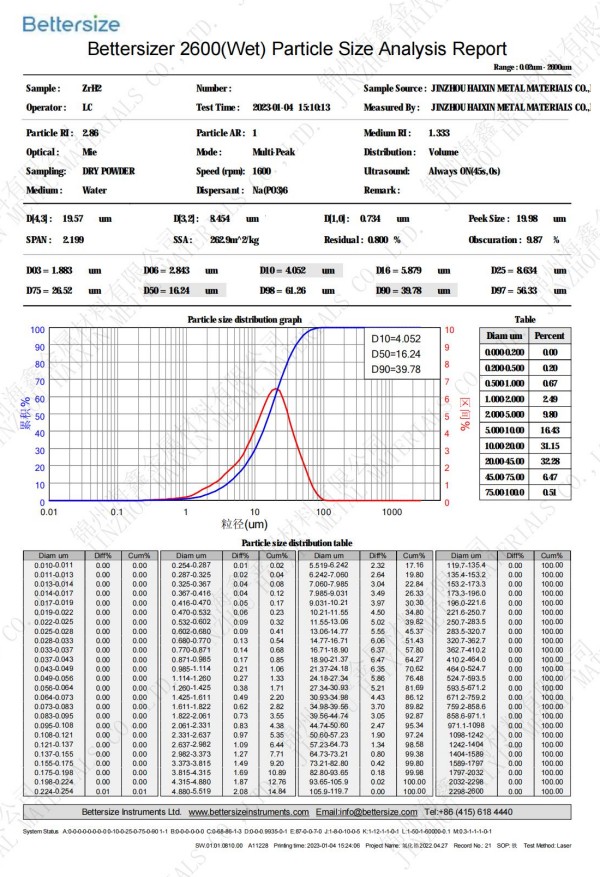





Titanium hydride (TiH2) is a metal hydride formed by the combination of titanium and hydrogen elements.
It is chemically active and needs to be stored away from high temperatures and strong oxidants.
Because it is relatively stable in air, it can also be used to prepare hydrogen gas and titanium hydroxide.
It can be obtained by directly reacting hydrogen gas with titanium metal.
At temperatures above 300 ℃, titanium metal can reversibly absorb hydrogen, ultimately forming a compound with the chemical formula TiH2.
If heated above 1000 ℃, titanium hydride will completely decompose into titanium and hydrogen gas.
At sufficiently high temperatures, titanium hydrogen alloys are in equilibrium with hydrogen gas, and the partial pressure of hydrogen gas is a function of the hydrogen content and temperature in the metal. Titanium hydride also has a complex hydrogen deficient phase with variable composition and different Ti-H spacing. These hydrogen deficient phases have been widely studied in recent years because they can be used to make buffers, reflectors, or high-temperature protective equipment, and may also be used in mobile nuclear reactors.
Application of titanium hydride
It can be used as a getter in the electro vacuum process, as a hydrogen source in the manufacture of foam metal, as a source of high-purity hydrogen, and also used to supply titanium to alloy powder in metal ceramic sealing and powder metallurgy.
Titanium hydride is very brittle and can be used to manufacture powder titanium. It is also used for welding. When titanium hydride is heated, it decomposes and releases new ecological hydrogen and metallic titanium, which can promote welding and increase weld strength. Can be used as a catalyst for polymerization reactions.
Technological innovation
Honesty is the foundation
Contact Number: +86-15698999555 |
Address: NO.6 ,SHENGHUA STREET,TAIHE DISTRICT, JINZHOU CITY, LIAONING PROVINCE, CHINA. |