
Product Name: Boron Powder (B)
Specification: 0.8-10um (D50)
Appearance: Irregular
Color: Black or Brown
Features: High strength, lightweight, high heat resistance
Application: Metallurgy, electronics, pharmaceuticals, ceramics, nuclear industry, chemical industry and other fields





Product Name: Boron Powder
Status: Powder
chemical formula: B
Particle size: 1-5 microns
Density: ρ=2.37g/cm3
Melting point: 2177-2301 ℃
Boiling point: 2550-2658 ℃
Crystal structure: amorphous, crystalline powder
Appearance: Light brown to grayish black metal powder
Physical and chemical characteristics:
Boron powder, also known as monomeric boron, has a high mass and volume calorific value. It is a light brown to grayish black powder with relatively active chemical properties, but relatively stable at room temperature. When the temperature reaches 300 degrees Celsius, it will be oxidized and ignited at a high degree, requiring 700 degrees Celsius to ignite.
Product Usage:
(1) Fireworks industry: Explosive triggers, boron flames appear green.
(2) Rocket and missile fuel rich propellants.
(3) Nuclear industry: neutron counter tubes, neutron absorbers, reactor control rods.
(4) Automotive airbags: initiators.
(5) Powder metallurgy additives, thermal spray surface coatings.
(6) Metallurgy: gas scavengers, smelting of special steel.
(7) Semiconductor industry: doping elements.
(8) Pharmaceutical industry and organic synthesis: catalysts.
(9) Synthetic borides: boron containing functional ceramics, boride superconductors, etc.
(10) The glass industry.
(11) Electronic industry: ignition electrode (cathode material) for ignition tubes.
(12) Welding powder: an important part of weapon welding.
matters needing attention:
△ Safety instructions: This product is toxic by inhalation or ingestion. Inhalation affects the central nervous system, ingestion can cause gastrointestinal irritation and boron poisoning, and skin and eye contact can cause mild irritation. When operating, wear safety goggles, dust masks, and rubber gloves.
Product packaging: vacuum packaging, aluminum platinum bag lined with plastic bag. Each bag has a net weight of 5kg.
Storage method: Store in a cool, ventilated, and dry warehouse. The shelf life is 12 months, and it can still be used after passing the re inspection.
Product transportation: During transportation, avoid severe collisions, rain, and direct sunlight, and do not mix with strong oxidants.
Attention: Elemental boron is a black or dark brown powder. When oxidized in air, the formation of boron trioxide film hinders the further oxidation of internal boron. Amorphous boron has active chemical properties. Powder and air can form explosive mixtures.
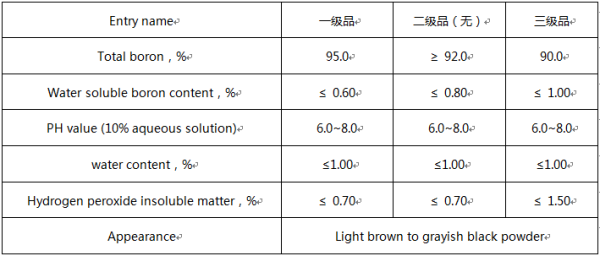
Technical index standard of boron powder
Technological innovation
Honesty is the foundation
Contact Number: +86-15698999555 |
Address: NO.6 ,SHENGHUA STREET,TAIHE DISTRICT, JINZHOU CITY, LIAONING PROVINCE, CHINA. |